VANADIS®: How it works in acute Myocardial Infarction
A heart attack (myocardial infarction) happens when a coronary artery is blocked, cutting off oxygen to part of the heart muscle. It’s a medical emergency and a major driver of mortality, cardiovascular disease is the leading cause of death worldwide. In the United States alone, every 40 seconds or roughly 2,100 times each day, a person has a heart attack, underscoring the urgency of rapid recognition and treatment.
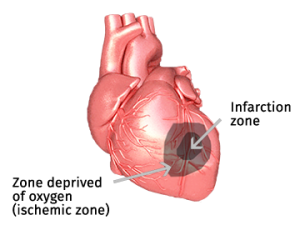
When a heart attack (myocardial infarction) occurs, a coronary artery becomes partially or completely blocked. Cells in the most deprived region die, this is the infarction zone. Surrounding it is the ischemic zone, where blood flow is severely reduced but cells remain viable. Because circulation is limited, metabolic waste is cleared poorly and accumulates inside these stressed cells.
Standard care is to reopen the artery with percutaneous coronary intervention (PCI), restoring blood flow. However, the sudden return of oxygen can trigger harmful chemical reactions with the built-up metabolites, causing additional cell death – reperfusion injury – and enlarging the infarct. VANADIS® is designed to blunt this injury. Ideally is it applied as early as possible following a myocardial infarction, e.g. at home or in the ambulance or around the time of PCI.
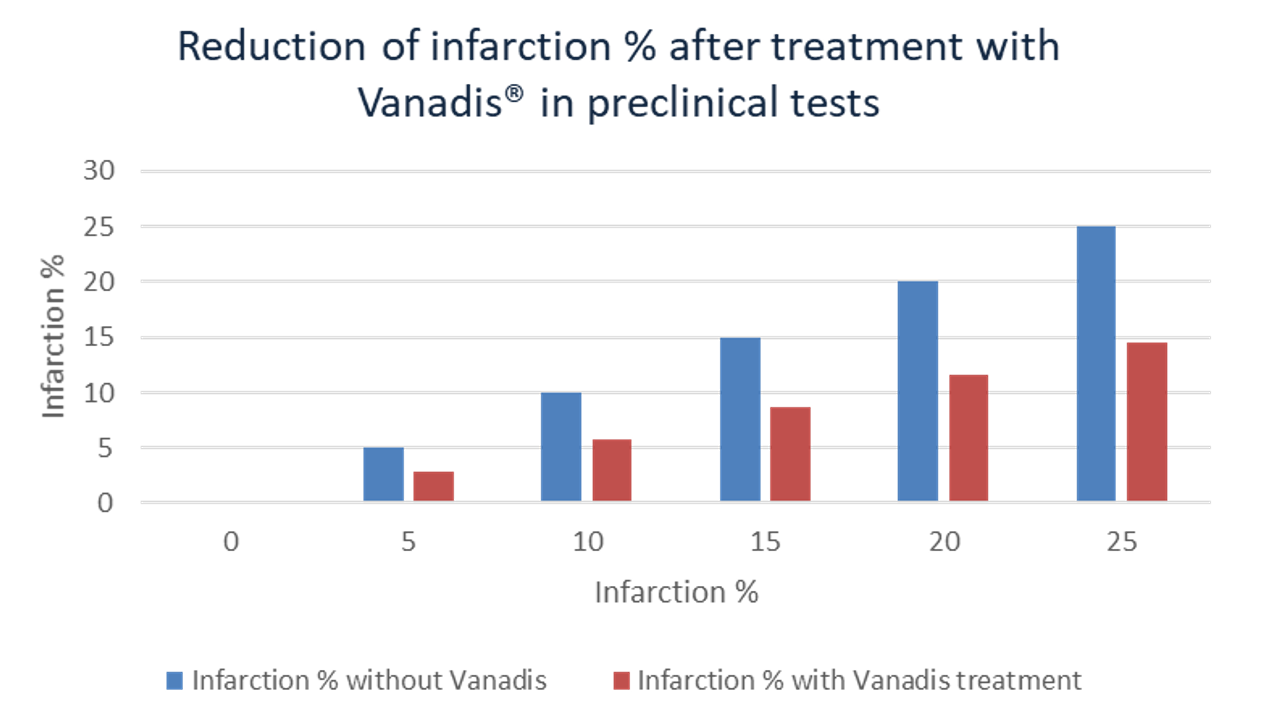
By making ischemic-zone cells more resilient to the abrupt oxygen surge, VANADIS® reduces cell death and limits the expansion of the infarct. In addition to its anti-apoptotic and pro-regenerative actions, VANADIS® targets multiple inflammatory and oxidative pathways that drive reperfusion damage. Preclinical studies show that early VANADIS® can cut infarct size by over 40%, which is expected to translate into a better heart pump function (see below) and, consequently, fewer complications such as recurrent ischemic events, heart failure, and mortality, with a better day-to-day quality of life.
What this can mean for patients
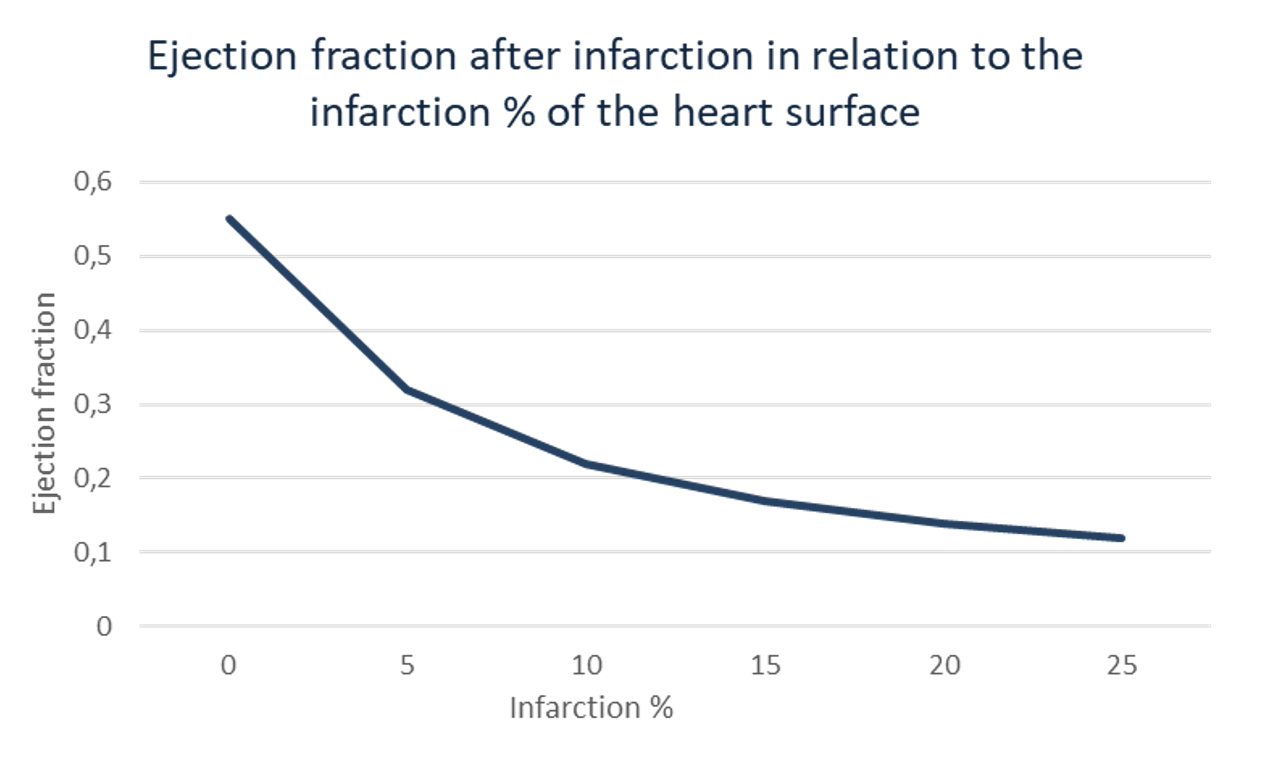
A heart attack weakens the heart’s pump function, commonly measured as the ejection fraction (EF), the percentage of blood the left ventricle ejects with each heartbeat. EF after an infarct is a major predictor of survival, quality of life, and downstream healthcare costs.
VANADIS® status and next steps
VANADIS® is administered intravenously and is intended to minimize ischemic and reperfusion damage and preserve heart function. On the strength of its mechanism of action, development has advanced from preclinical work to clinical testing.
-
Preclinical: Completed with strong signals, including >40% infarct-size reduction and pro-repair effects
-
Phase 1A (healthy volunteers): Completed; VANADIS® was well tolerated with no serious adverse events at therapeutic levels
-
Planned clinical program: Phase 1B and Phase 2A trials in acute Myocardial Infarction to assess:
-
Reduction infarct size
-
Improved heart performance (ejection fraction)
-
Lowered risk of heart failure
-
Reduced mortality
-
Protection of other organs (e.g., kidneys and brain) from secondary damage due to heart failure
-
Upon successful completion of Phase 2A trials for Myocardial Infarction, CFM Pharma intends to partner with one of the leading pharmaceutical companies in cardiovascular disease. In close collaboration with Key Opinion Leaders and University Medical Centers the CFM Team is committed to translating VANADIS® into real-world patient care as fast as possible.
