The compound
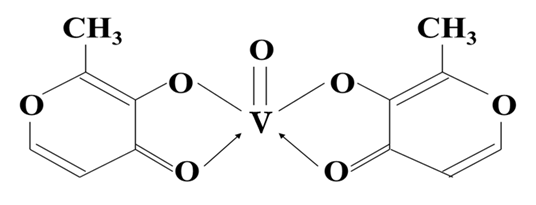
CFM has a platform of organovanadium compounds and delivery technologies of which the first one is entering into clinical phase II trials in several trauma indications. This compound, BMOV (bis(maltolato)oxidovanadium (IV)) is an organic molecule:
Effect of VANADIS® on cells
VANADIS® has an effective protective mechanism that helps to keep traumatized (but still) healthy cells alive and that stimulates repair of damaged tissue. Preclinical research results have demonstrated this positive effect for multiple trauma indications.
Application area’s of VANADIS®
CFM Pharma has a number of important applications of VANADIS® under development:
- Treatment of patients with myocardial infarction. Preclinical studies have shown that the size of the infarction can be limited significantly.
- Treatment of deep skin burns, diabetic and general wounds. Preclinical studies show that the affected tissue regenerates better and that up to 90% reduction in tissue damage can be achieved.
- Treatment of other trauma indications like acute kidney injury. Preclinical studies have shown that VANADIS® can limit reperfusion injury in kidneys during acute renal failure.
Given the broad applicability of VANADIS® in trauma conditions, more indications are expected to be developed in the future, such as treatment of brain infarcts (stroke).
VANADIS® can be used safely
The organic vanadium compound BMOV (VANADIS®), has been shown to be safe and well tolerated in the concentrations applied due to its long half-life (it is known that some vanadium compounds can be toxic with prolonged use and high doses).
The preclinical and phase 1 studies performed with volunteers have shown that for the applied doses, BMOV is safe for further drug development in traumatic conditions such as myocardial infarction and burn wounds.
